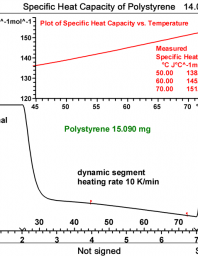
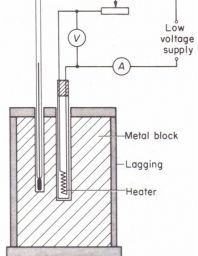
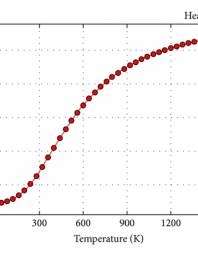
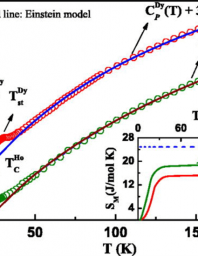
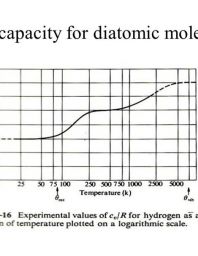
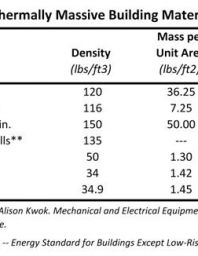
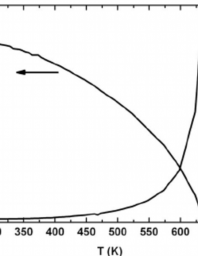
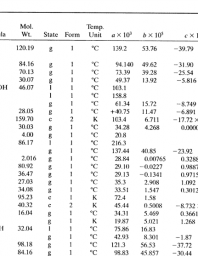
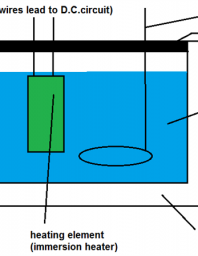
Heat capacity determination
Heat capacity – the ratio of the energy to temperature. It is the physical quantity that shows the amount of the heat must be used order to change its temperature by one unit K or °C. This amount of energy can be different (but not always positive) and depends on the conditions and definite substance.
Specific heat capacity for different materials
Specific heat capacity – the ratio of heat capacity to mass, heat capacity of a unit mass of the substance (different for different substances). A physical quantity that is equal of the amount of the heat that must be transferred to the unit mass of the substance in order to change its temperature by one unit K or °C.
The more thermal energy is wasted to each gram, the faster temperature change. The specific heats of any materials are different. Therefore, you need to know how to identify this quantity.
In general, the specific heat capacity is the ability of a medium to store heat per unit rise in the temperature of the medium and per unit mass.
Formulation of specific heat capacity
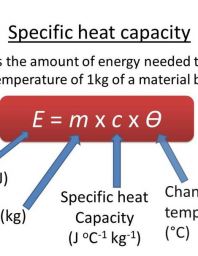
To calculate specific heat, you use the following equation:
Where:
- Q – the amount of the energy
- c – specific heat of capacity
- m – the mass of the substance or material
- ∆T – temperature changing of the substance during the heating
Types of substances of a specific heat capacity
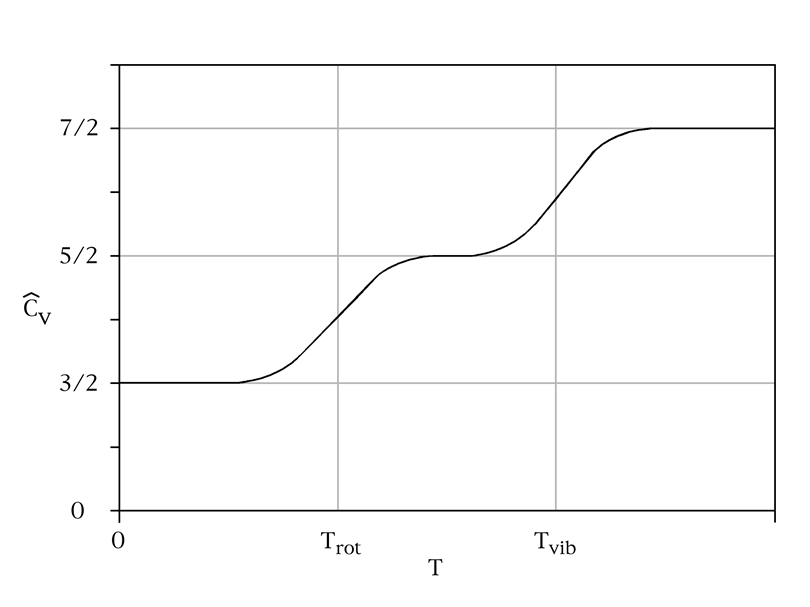
Substances exist in three forms: solid, liquid and gaseous state. The main difference between all these stats is how the molecules are located and what connections between them, it shows the spreading the heat in the substance. And only water can appear in all three forms as water, ice, and steam.
- In solid state: the heat, due to close connection of elements in crystal cell, spreads faster than in any other form.
- In gaseous state: though the distant between the molecules is far, the heat are spreading via the banding of molecules, they star to move quickly and transport the heat to others. It also depends on pressure in gas.
- In liquid state: the molecules are not very closely and not very far from each other. Therefore, the molecules in liquid require much more time than any other substances and liquid with the highest specific heat.
Specific heat capacity table of common materials
When it comes to measure the specific heat capacity for a particular substance, we have the possibility to use have already measured quantities, which are picked out in a special table of specific heats of various substances. Here the table of some specific heats of common materials at room temperature:
| Substance | Specific Heat | |
| J/kg * K | Cal/g * °C | |
| Water | 4180 | 1 |
| Air | 1001 | 0.2391 |
| Styrofoam | 1131 | 0.2701 |
| Copper | 386 | 0.0923 |
| Glass | 840 | 0.20 |
| Lead | 128 | 0.0305 |
| Steel | 500 | 0.107 |
| Aluminum | 897 | 0.902 |